Introduction
Have you ever wondered how drugs get their names? It’s not a random process but a well-structured system known as drug nomenclature. This article will take you on a journey through the intricate world of drug naming, shedding light on the logic and science behind the names on your medicine bottles.
Drug Nomenclature: Drug Naming System
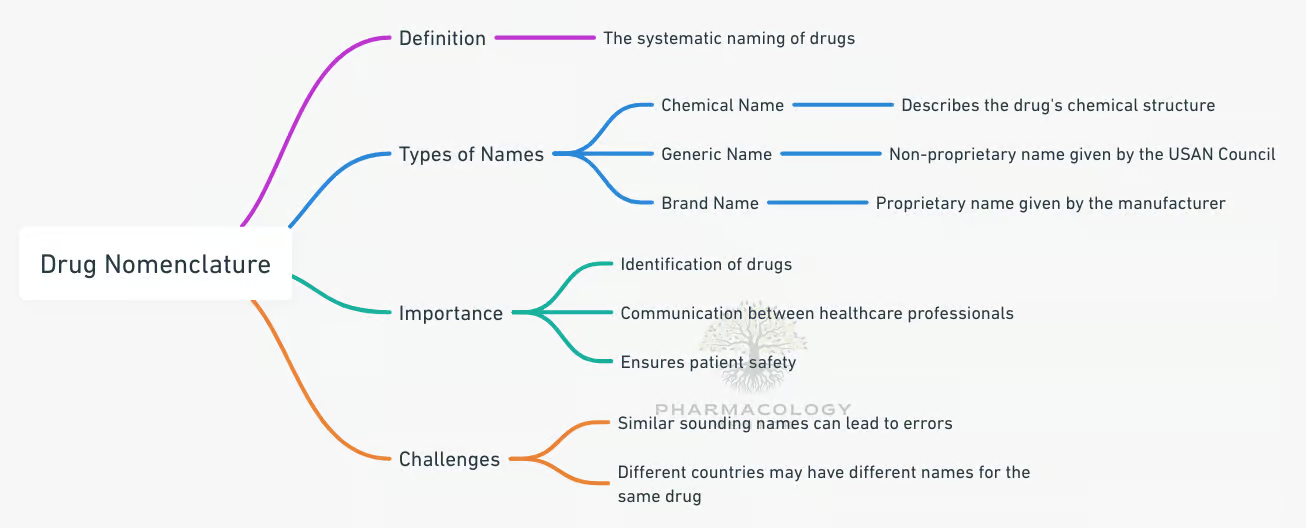
Drug nomenclature is a systematic approach to naming drugs. It’s not as simple as picking a name out of a hat; it’s a complex process that involves scientists, researchers, and regulatory bodies from around the world. Let’s delve deeper into this fascinating system.
The Science Behind Drug Names
Drug names are not just random combinations of letters. They are carefully crafted to convey specific information about the drug. The name can tell you about the drug’s chemical structure, its pharmacological properties, and its therapeutic category. It’s like a mini science lesson in every name!
The Art of Naming Drugs
While there is a lot of science involved in drug naming, there’s also an art to it. The name needs to be unique, easy to pronounce, and memorable. It’s a delicate balancing act between scientific accuracy and marketing appeal.
The Role of Regulatory Bodies
Regulatory bodies like the U.S. Food and Drug Administration (FDA) and the World Health Organization (WHO) play a crucial role in the drug naming process. They review proposed drug names for safety and potential confusion with existing drug names. Their goal is to ensure that drug names are safe, effective, and user-friendly.
The Anatomy of a Drug Name
Drug names may seem like a jumble of letters, but there’s a method to the madness. Each part of a drug name has a specific meaning and purpose.
Chemical Name
The chemical name of a drug describes the substance chemically. It is assigned according to chemical nomenclature rules and is often very complex and cumbersome. For example, propranolol’s chemical name is 1-(Isopropylamino)-3-(1-naphthyloxy)propan-2-ol, which is unsuitable for prescribing. To make it more convenient and simple, the manufacturer may assign a code name for the drug, such as RO 15-1788, before an approved name is coined.
Non-Proprietary Name
The non-proprietary name of a drug is the name accepted by a competent scientific body or authority, such as the USAN or BAN. The non-proprietary names of newer drugs are kept uniform by an agreement to use the Recommended International Nonproprietary Name (rINN) in all member countries of the World Health Organization (WHO). The BAN of older drugs has also been modified to be commensurate with rINN. However, many older drugs still have more than one non-proprietary name. For example, meperidine and pethidine, and lidocaine and lignocaine are the same drugs with different non-proprietary names. Until the drug is included in a pharmacopoeia, the non-proprietary name may also be called the approved name. After its appearance in the official publication, it becomes the official name.
In common parlance, the term generic name is used in place of a nonproprietary name. Etymologically, this is incorrect. Generic should be applied to the chemical or pharmacological group (or genus) of the compound, such as phenothiazines, tricyclic antidepressants, and aminoglycoside antibiotics. However, this misnomer is widely accepted and used even in official parlance.
Proprietary (Brand) Name
The proprietary (brand) name of a drug is assigned by the manufacturer and is his property or trademark. One drug may have multiple proprietary names from different manufacturers. For example, amlodipine has multiple proprietary names, such as AMCARD, AMLOGARD, AMLOCOR, AMLONG, AMLOPIN, AMLOVAS, and STAMLO. Brand names are designed to be catchy, short, easy to remember, and often suggestive. For example, LOPRESOR suggests a drug for lowering blood pressure. Brand names generally differ in different countries. For example, timolol maleate eye drops are marketed as TIMOPTIC in the USA but as GLUCOMOL in India. Even the same manufacturer may market the same drug under different brand names in different countries. In addition, combined formulations have their own multiple brand names. This is responsible for much confusion in drug nomenclature.
Advantages of Using Non-Proprietary Names in Prescribing There are many arguments for using the non-proprietary name in prescribing.
The Process of Naming a Drug
Naming a drug is a long and complex process that involves many steps. Let’s take a closer look at how it’s done.
Chemical Discovery and Naming
The process begins when a new drug molecule is discovered. The scientists who discover the molecule give it a chemical name based on its molecular structure.
Development of the Generic Name
Next, a generic name is developed. This is done by a group of experts who follow specific guidelines to ensure that the name is informative, unique, and easy to pronounce.
Brand Name Creation
The last step is the creation of the brand name. This is done by the drug manufacturer, who must come up with a name that is catchy, memorable, and does not infringe on any existing trademarks.
Challenges in Drug Naming
Despite the systematic approach to drug naming, there are still challenges that need to be addressed.
Avoiding Confusion
One of the biggest challenges in drug naming is avoiding confusion with existing drug names. This is crucial for patient safety, as confusion can lead to medication errors.
Global Consistency
Another challenge is ensuring global consistency in drug names. With so many different languages and alphabets, it can be difficult to come up with a name that is universally understood and pronounced.
Trademark Issues
Trademark issues can also pose a challenge. Drug manufacturers need to ensure that their brand name does not infringe on any existing trademarks.
The Future of Drug Naming
The world of drug naming is constantly evolving. As new drugs are discovered and new naming guidelines are developed, the drug naming system will continue to adapt and improve.
Innovation in Drug Naming
Innovation is key in the world of drug naming. From using artificial intelligence to generate unique drug names to developing new naming conventions for gene therapies, the future of drug naming is full of exciting possibilities.
Improving Patient Safety
Improving patient safety is a top priority in the future of drug naming. This includes developing strategies to reduce confusion between drug names and improving the readability and understandability of drug names.
Global Harmonization
Global harmonization of drug names is another important goal for the future. This involves working towards a unified global system for drug naming to ensure consistency and reduce confusion.
Conclusion
Drug nomenclature is a fascinating blend of science, art, and regulation. It’s a complex system that plays a crucial role in our healthcare system. By understanding the drug naming system, we can better understand the medicines we use and the world of pharmaceuticals that surrounds us.
Disclaimer: This article is for informational purposes only and should not be taken as medical advice. Always consult with a healthcare professional before making any decisions related to medication or treatment.
FAQs
1. What is drug nomenclature?
Drug nomenclature is the systematic approach to naming drugs. It involves a combination of science, art, and regulatory oversight to ensure that drug names are informative, unique, and user-friendly.
2. What are the parts of a drug name?
A drug name typically consists of a chemical name, a generic name, and a brand name. The chemical name describes the drug’s molecular structure, the generic name is a simplified version of the chemical name, and the brand name is the name given to the drug by its manufacturer.
3. How is a drug named?
Naming a drug is a complex process that begins with the discovery of a new drug molecule. The molecule is given a chemical name, then a generic name is developed, and finally, the drug manufacturer creates a brand name.
4. What challenges are there in drug naming?
Some of the challenges in drug naming include avoiding confusion with existing drug names, ensuring global consistency in drug names, and dealing with trademark issues.
5. What is the future of drug naming?
The future of drug naming involves innovation in naming practices, improving patient safety, and working towards global harmonization of drug names.
6. Why is drug naming important?
Drug naming is important because it provides a standardized way to identify drugs. It helps healthcare professionals communicate about drugs and helps patients understand what they are taking.